Nootropic Properties of Huperzia Serrata
Nootropic Properties of Huperzia Serrata
Background
Huperzia Serrata is a species of fir clubmoss containing a biologically active compound known as Huperzine A (HupA). This herbal extract has a well-characterized inhibitory effect on acetylcholinesterase (AchE), an enzyme responsible for degrading the neurotransmitter acetylcholine (Ach). Efficient Ach neurotransmission is critical in processes underlying learning, memory, and attention. Reduced levels of Ach neurotransmission are hypothesized to contribute to cognitive decline and memory impairments associated with age and neurological diseases. The use of HupA as a novel treatment for memory impairments has shown promising results. Furthermore, HupA supplements have produced enhanced attention and memory in healthy subjects. Further studies on animal models show the biological efficacy of HupA on cholinergic neurons, as well as corroborating evidence of improvement in cognitive skills.
Significant Findings
Huperzine A boosts acetylcholine and prevents oxidative stress in cholinergic neurons.
HupA is a highly specific reversible inhibitor of AchE, an enzyme responsible for the removal of Ach in the synaptic cleft of cholinergic neurons. HupA efficiently crosses the blood-brain barrier, rapidly binding to the active site residues to form the Ache-HupA complex [1]. Although structural analysis of HupA bears no resemblance to the neurotransmitter Ach, it has higher binding affinity than other bioengineered AchE inhibitors and displays a slower rate of dissociation [2]. A study of the pharmokinetics of HupA administration in healthy human volunteers reported a peak plasma concentration after 58.33±3.89 minutes, with an elimination half-life of 716.25 ± 130.18 minutes (Fig 1)[3].
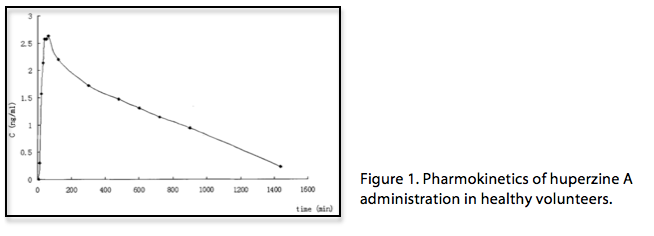
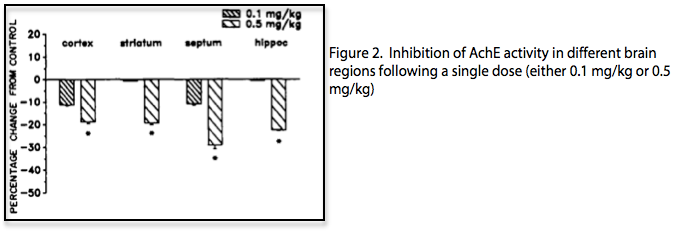
Both in vitro and in vivo studies of HupA highlight the specificity of the inhibitor and its effect on neurotransmitter levels. In cortical cells, prolonged incubation of HupA did not decrease the functionality of AchE, and the activity of AchE returned to 94% of baseline with subsequent washes, demonstrating its reversibility. Oral administration of HupA caused significant increases in Ach concentration in animal brains. These increases were regionally selective and showed specific increases in the cortex and hippocampus as early as 30 minutes after administration [4]. Given that cortical Ach is low in Alzheimer’s disease (AD), this specific binding of HupA presents a possible therapeutic advantage. A single dose (0.5 mg/kg) is sufficient to significantly inhibit AchE in important brain regions (Fig 2)[5]. Repeated doses of HupA show no decline of AchE inhibition compared to a single dose, indicating no development of tolerance with HupA administration [2]. Preclinical studies using animal models showed that HupA administration ameliorates learning and memory deficits through its inhibitory effects [6].
HupA has demonstrated additional neuroprotective properties beyond its inhibitory role. Apoptosis, or programmed cell death, through production of a precursor peptide known as amyloid beta, is the central working hypothesis for the cognitive decline in AD. HupA has been shown to protect neurons against apoptosis in amyloid beta stress models. The precise anti-oxidative mechanism of HupA has recently been explored. The cellular response to amyloid production generates free radicals, which in turn damage mitochondrial enzymes, leading to impaired glucose metabolism and eventually neuronal death. HupA exhibits cytoprotective effects on key mitochondrial components, reducing neuronal apoptosis and suggesting a possible role in the prevention and treatment of neurodegenerative disease [7].
Huperzine A improves cognition in patients with memory deficits
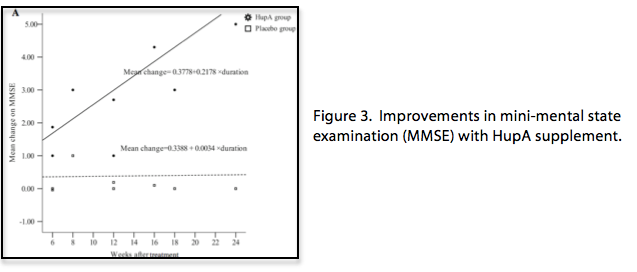
The historical use of HupA and discovery of its molecular activity have led researchers to investigate HupA as a therapy for cognitive disorders and neurodegenerative disease. Clinical trials evaluating cognition in AD show statistically significant memory improvements with HupA administration compared to a placebo. A study in 2002 enrolled over two hundred patients meeting the criteria for AD. The patients who were given HupA showed significant improvements in cognitive function, such as orientation, attention and memory. In addition, non-cognitive evaluations, such as mood and behavior, improved after both 6 and 12 weeks of administration. A recent meta-analysis highlights the memory improvements with HupA treatment, correlating even greater improvements with prolonged use (Figure 3)[8]. The percentage of mild and adverse side effects were low (3%), indicating that HupA is safe and effective over a 12-week period.
A similar study examined HupA in treating vascular dementia, one of the most common causes of dementia affecting the quality of life for both patients and their families. The study reported significant improvements in cognitive functions such as memory, calculation, language and visual spatial abilities after 8 weeks of treatment [9]. Furthermore, HupA was well tolerated and patients experienced minimal adverse side-effects.
Huperzine A enhances memory in non-patient populations
A 1999 study in China aimed to evaluate the memory enhancing effects of HupA in adolescent students. 34 pairs of students who complained of memory inadequacy were administered HupA or placebo orally. After 4 weeks of administration, evaluation using the Welschler memory scale showed significant improvements in accumulation, recognition, reproduction, association, tactual memory, and recitation. In addition, there were significant improvements in Chinese and English language quizzes and the study reported no side effects [10]. These results suggest HupA as a promising candidate for improving memory function in healthy individuals.
Summary
Extensive research has revealed the biological mechanism of HupA, most notably its inhibition of AchE and secondary neuroprotective effects. Preclinical studies in animals provide evidence of increased Ach neurotransmitter concentration in cognitively important brain regions along with learning and memory improvements. These preclinical studies and the historical use of Huperzia Serrata as a memory supplement led researchers to investigate the possible benefits in cognitive disorders. Double-blind, placebo controlled studies have shown that Alzheimer’s and vascular dementia patients exhibit significant improvements in memory function after HupA treatment. Furthermore, non-patient populations display similarly enhanced memory function with oral HupA supplement. The low incidence of adverse side effects allows Huperzia Serrata and Huperzine A extracts to be used safely as nootropic supplements to support memory and neurological health.
Reference
- Ashani Y, Peggins JO, 3rd, Doctor BP: Mechanism of inhibition of cholinesterases by huperzine A. Biochem Biophys Res Commun 1992, 184:719-726.
- Zangara A: The psychopharmacology of huperzine A: an alkaloid with cognitive enhancing and neuroprotective properties of interest in the treatment of Alzheimer’s disease. Pharmacol Biochem Behav 2003, 75:675-686.
- Li YX, Zhang RQ, Li CR, Jiang XH: Pharmacokinetics of huperzine A following oral administration to human volunteers. Eur J Drug Metab Pharmacokinet 2007, 32:183-187.
- Tang XC, De Sarno P, Sugaya K, Giacobini E: Effect of huperzine A, a new cholinesterase inhibitor, on the central cholinergic system of the rat. J Neurosci Res 1989, 24:276-285.
- Laganiere S, Corey J, Tang XC, Wulfert E, Hanin I: Acute and chronic studies with the anticholinesterase Huperzine A: effect on central nervous system cholinergic parameters. Neuropharmacology 1991, 30:763-768.
- Cheng DH, Tang XC: Comparative studies of huperzine A, E2020, and tacrine on behavior and cholinesterase activities. Pharmacol Biochem Behav 1998, 60:377-386.
- Gao X, Tang XC: Huperzine A attenuates mitochondrial dysfunction in beta-amyloid-treated PC12 cells by reducing oxygen free radicals accumulation and improving mitochondrial energy metabolism. J Neurosci Res 2006, 83:1048-1057.
- Wang BS, Wang H, Wei ZH, Song YY, Zhang L, Chen HZ: Efficacy and safety of natural acetylcholinesterase inhibitor huperzine A in the treatment of Alzheimer’s disease: an updated meta-analysis. J Neural Transm 2009, 116:457-465.
- Xu ZQ, Liang XM, Juan W, Zhang YF, Zhu CX, Jiang XJ: Treatment with huperzine a improves cognition in vascular dementia patients. Cell Biochem Biophys, 62:55-58.
- Sun QQ, Xu SS, Pan JL, Guo HM, Cao WQ: Huperzine-A capsules enhance memory and learning performance in 34 pairs of matched adolescent students. Zhongguo Yao Li Xue Bao 1999, 20:601-603.